
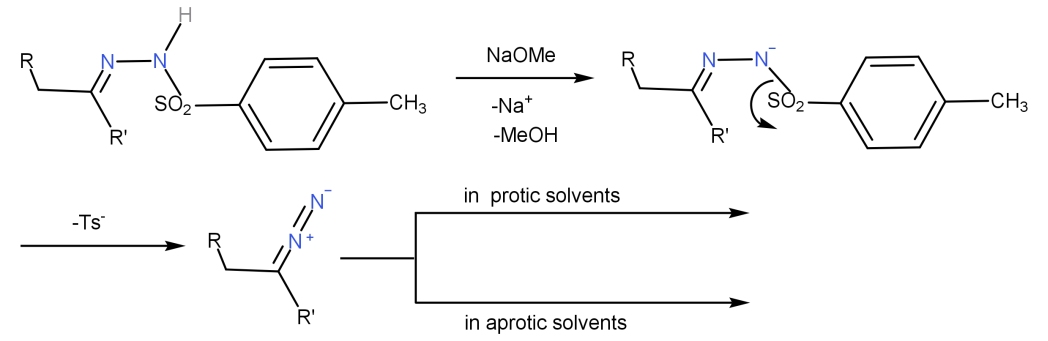
Highly efficient methodology for the reductive coupling of aldehyde tosylhydrazones with alkyllithium reagents. Reductive alkylation of aldehyde tosylhydrazones with organolithium reagents. N-Tosylhydrazones as reagents for cross-coupling reactions: a route to polysubstituted olefins. Stereoselective synthesis of cis-disubstituted oxygen and nitrogen heterocycles. Ruthenium porphyrin catalyzed intramolecular carbenoid C-H insertion. Application of chiral sulfides to catalytic asymmetric aziridination and cyclopropanation with in situ generation of the diazo compound. K., Alonso, E., Fang, G., Ferrara, M., Hynd, G. Catalytic asymmetric synthesis of epoxides from aldehydes using sulfur ylides with in situ generation of diazocompounds. A novel route to 2-arylcyclopropylamines. Catalytic cyclopropanation of alkenes using diazo compounds generated in situ. The use of tosylhydrazone salts as a safe alternative for handling diazo compounds and their applications in organic synthesis. Dual labeling of biomolecules by using click chemistry: a sequential approach.

Click chemistry and medicinal chemistry: a case of ‘cyclo-addition’. Click chemistry: diverse chemical function from a few good reactions. Domino iron catalysis: direct aryl–alkyl cross-coupling. Green Chemistry and Catalysis (Wiley-VCH, 2007).Ĭzaplik, W. Metal-Catalyzed Cross-Coupling Reactions (Wiley-VCH, 2004). As the required tosylhydrazones are easily generated from carbonyl compounds, it can be seen as a reductive coupling of carbonyls, a process of high synthetic relevance that requires several steps using other methodologies.ĭe Mejiere, A. This reaction is very general and functional-group tolerant. Here, we report a new and efficient metal-free carbon–carbon bond-forming coupling between tosylhydrazones and boronic acids. Similarly, efficient processes that do not require a metal catalyst are of extraordinary interest. In this context, the development of new cross-coupling reactions that use catalysts based on inexpensive and non-toxic metals is attracting increasing attention. In spite of the myriad methods available, advantageous methodologies in terms of selectivity, availability of starting materials, operational simplicity, functional-group tolerance, environmental sustainability and economy are in constant demand. The formation of carbon–carbon bonds is a fundamental transformation in organic synthesis.

